Demonstration experiments
Increase in volume through the formation of gases
If unexpectedly large amounts of gases or vapours are released during reactions, this may lead to an increase in pressure that an apparatus can no longer contain. In a model experiment, a hydrogen peroxide solution and a potassium iodide solution, which catalyse the breakdown of H2O2, are simultaneously added to a small amount of liquid detergent. A mountain of suds immediately forms, vividly showing how splitting off oxygen leads to an increase in volume.
You can download the video clip here:
Absorption and desorption of gases by respiratory filters
Dried activated charcoal is added via an insertion device to the round-bottomed flask of a closed apparatus that has been filled with chlorine gas. To achieve better contact between the solid surface of the activated charcoal and the gaseous phase, a magnetic stirrer is used to stir the activated charcoal as it is added. On the right side, the flask is connected to a second, air-filled flask via a Teflon tube, which in turn is connected to an Erlenmeyer flask in the middle containing a dip bath of dyed blue water. You can see that the blue water is pushed back into the air-filled flask as the gas pressure decreases in the first flask through the rapid adsorption of the chlorine to the activated charcoal.
Once the activated charcoal is saturated with chlorine, it can be desorbed again by means of heating with a hot air dryer. This pushes the dyed blue water (the dye slowly fades in reaction to chlorine) back into the round-bottomed flask on the right side.
Dried activated charcoal is added a second time while stirring (the excess is held back in the absorption tower to the far right), and chlorine is then passed over until the activated charcoal is saturated with chlorine. This activated charcoal is then outgassed under vacuum in a round-bottomed flask and the desorbed chlorine gas is frozen in a nitrogen cold trap. As the trap thaws, it can be seen that the chlorine that had been absorbed by the activated charcoal has melted and is already boiling.
In another experiment, the activated charcoal is exposed to water vapour for a prolonged period of time. If chlorine is then allowed to act upon the charcoal before being transported into the cold trap through outgassing, it will be found to contain practically only water; the moist activated charcoal will not have been able to absorb the chlorine.
Respiratory filters only have a limited capacity of absorbing hazardous gases and vapours; any excess permeates the filter. Only respiratory filters that have been stored properly offer sufficient protection.
You can download the video clip here:
An experiment on the protective function of a laboratory coat
Sugar cubes are placed in three watch glasses. Either a piece of fabric from a laboratory coat or a piece of a conventional clothing fabric is placed on two of the cubes. A few drops of concentrated sulfuric acid are added to all three samples. The acid rapidly breaks down the sugar to black coal-like reaction products as water is split off and sulphuric acid hydrates are produced exothermically.
A hole soon forms in the clothing fabric, burned into the fabric by the acid; the sugar underneath is also affected. The piece of laboratory coat withstands the acid for a much longer period of time; a small amount of sulfuric acid penetrates, leading to a discolouration of the sugar. This clearly demonstrates the protective function of a laboratory coat, which can at times be a surprisingly effective barrier against contamination.
You can download the video clip here:
Breakthrough behaviour of glove material
You believe that you are protected after you have put on your personal protective equipment. And this is in fact the case if the PPE has been correctly and appropriately selected for the task at hand and is used properly. Otherwise, it may provide a dangerous, false sense of security, the consequences of which may be detrimental to health or even fatal.
Choosing the right glove requires expert knowledge. Always choosing the same glove, just because it is the one that has always been used and the one – perhaps even the only one – that is available, may have dangerous consequences.
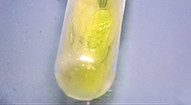
In the experiment, the finger of a nitrile disposable glove is cut off and dipped into an alkaline potassium triiodide solution, 1 ml acetone is added using a pipette and the finger is sealed at the top. Within a few seconds, the yellow streaks of a triiodomethane precipitate (iodoform test) begin to form on the glove surface, which shows that acetone is penetrating the material of the glove.
This demonstrates just how quickly a fairly common solvent like acetone (and quite a few others) can permeate the material of an incorrectly chosen glove and, in doing so, transport dissolved substances through to the skin. The acetone example also reveals another problem: practically no glove material can resist the permeation of acetone for prolonged periods of time.
You can download the video clip here:
Chemical destruction of gloves
The finger of a nitrile disposable glove is cut off and filled with anhydrous nitric acid. The glove spontaneously ignites because of the high oxidative power of nitric acid. None of the currently available gloves can guarantee safe work in this instance. Instead, the procedure should be designed in such a way that contact with the hand is avoided. In the experiment shown, this is achieved by using a stand clamp of sufficient length.
You can download the video clip here:
An experiment on the flammability of glove material
Approximately 2 ml of a tert-butyllithium solution (1.9 m in pentane) are sprayed on a nitrile disposable glove and on a Viton glove. Within just a few seconds, the pyrophoric liquid spontaneously ignites and burns the nitrile glove down to a little dry ash. The Viton glove does not ignite, but decomposes in the flames and tears apart. Thus, when assessing a hazard, it must be carefully considered whether the hazard to the hands resulting from contact with the pyrophoric liquid is greater than the hazard arising from a burning glove that can only be removed from the hand with difficulty. An important factor is the safe execution of all operations necessary to carry out the chosen procedure to avoid any additional hazards caused by clumsiness resulting from the reduced tactility inherent to wearing especially thick gloves.
You can download the video clip here:
Hydrofluoric acid destroys glass
Materials that are not resistant to chemical and physical impacts will cause apparatus and systems to fail, which may sometimes have very serious consequences.
In the experiment, a small amount of 40 per cent hydrofluoric acid is added to a small, thin-walled glass flask made of borosilicate glass 3.3, the glass commonly used for laboratory equipment and glass apparatus. The wall of the flask is completely destroyed within a few minutes and hydrofluoric acid escapes.
You can download the video clip here:
Reaction of iodine with chlorine
Many oxidation processes release a large amount of heat. This not only involves the ignition of substances exposed to (mainly atmospheric) oxygen; oxidation processes also occur between substances that would normally not be expected to react in such a way. In the experiment, chlorine acts as an oxidant in the reaction with elemental iodine. Iodine is spontaneously oxidized to iodine monochloride and, if there is an excess of chlorine, also partially to iodine trichloride. As soon as chlorine is introduced into the reaction vessel, iodine monochloride forms red-brown vapours that condense to a liquid.
Although most combustion reactions that cause damage and accidents involve a reaction with atmospheric oxygen, a risk assessment also needs to take other reactions into account.
You can download the video clip here:
Reaction of chlorine with copper and iron
Chlorine is a strong oxidant; many substances will burn just as vigorously in chlorine as in oxygen. The two experiments show how violently copper (here: copper wires) and iron (here: steel wool) react with a stream of chlorine gas. The smoke that forms is made up of the anhydrous chlorides of the metals.
You can download the video clip here:
Reaction of aluminum with iodine
A large number of substances react only under certain conditions such as temperature or pressure. These kinds of processes may harbour hidden dangers if onset is delayed or initiated by contact with an extinguishing or cleaning agent. In the experiment, a little amount of water is added to a mixture of aluminum and iodine, wetting it slightly as is common practice for binding dust to remove dust deposits. In this case, however, this dust-binding method, though well-intentioned, leads to a vigorous reaction that generates high levels of heat and releases copious amounts of the violet vapour of corrosive iodine.
If, once the residue has cooled and is ready for disposal, water were added again, heat would be generated and lead to the formation of corrosive hydrogen iodide from the anhydrous aluminum(III) iodide that has been produced. Therefore, the safety data sheet information on hazardous reactions must also be observed during cleaning and disposal.
Reaction of potassium with water
The metals in Group 1 of the Periodic Table of Elements are very reactive substances. Their reactivity increases with atomic number, from lithium to sodium and potassium to rubidium and caesium. Although the first three substances can be stored under a protective liquid (mineral oil) to protect them from reacting with the air (however, the metals age at different rates to form hazardous reaction products; see also "Dangers caused by alkali metal stocks" and T. H. Brock, A. Ahrens-Moritz, D. Reichard: Nachrichten aus Chemie, Technik und Laboratorium 46 (1998) 16), rubidium and caesium need to be stored in a controlled atmosphere in sealed ampoules.
In the experiment, oil and crusts are removed from a small amount of potassium (see the references above for a discussion of the problem of the crusts), which is then placed into a beaker with water and the indicator phenolphthalein. The potassium reacts spontaneously to form hydrogen, which ignites immediately. After a short time, the molten potassium ball explodes due to the accumulation of an ever-increasing positive charge. Electrostatic repulsive forces that act between the positive charges eventually tear the ball apart. In addition to the hot potassium hydroxide solution that has been formed, this propels glowing potassium and potassium hydroxide particles in every direction. If the piece of potassium that is used is just a little too large, this will lead to a violent explosion that is extremely difficult to control.
At the end of the video, snapshots of the potassium, rubidium, lithium and sodium metals (clockwise from top left) are shown as they come into contact with water.
The experiment demonstrates the high reactivity of a number of substances and shows that water, the most widely used extinguishing agent, may be incompatible with certain substances.
You can download the video clip here:
Magnesium burns in carbon dioxide
Carbon dioxide is widely used as a fire-extinguishing agent (“carbon dioxide extinguisher”) and can put out many types of fires. However, an additional hazard may arise if the burning substances react with the fire-extinguishing agent.
In the experiment, magnesium chips (for Grignard reactions) are ignited in a block of dry ice (commercial solid carbon dioxide at a temperature of –78°C), and a second block of dry ice is placed on top of the first block to extinguish the fire. The cold extinguishing agent does not put out the fire, but accelerates it. As the magnesium burns, it snatches oxygen from the carbon dioxide molecules to form magnesium oxide (which produces the glaring white glow), releasing large amounts of heat. The product of the reaction is a mixture of white magnesium oxide and black carbon from the carbon dioxide.
One factor that must always be considered when developing a fire (or damage) response plan is the possibility of hazardous reactions.
You can download the video clip here:
You can reach the German-language version of the article with a single click on the flag-symbol.